Amino-modified oligonucleotides are synthetic DNA or RNA molecules containing primary amine functional groups
that enable covalent conjugation to dyes, ligands, surfaces, proteins, polymers, nanoparticles, and other functional materials.
These amine handles are among the most versatile reactive groups used in post-synthetic oligo labeling and bioconjugation workflows.
Amino modifiers can be introduced at the 5′ terminus, 3′ terminus, or internal positions using terminal linker phosphoramidites, amino-functionalized nucleobases, aminoallyl nucleotides, PEG-based spacers, and specialized functional modifiers such as aminooxy or photocleavable amino linkers. These modifications support probe labeling, microarray immobilization, biosensor construction, antibody–oligo conjugates, nanoparticle attachment, and broader macromolecule conjugation strategies.
Amino-modified oligos are widely used for covalent surface attachment in microarrays, biosensors, and bead-based capture systems, where spacer length can strongly influence accessibility, coupling efficiency, and downstream analytical performance.
Primary amine functional groups introduced through amino linkers are among the most widely used chemical handles in oligonucleotide bioconjugation. These amines readily react with N-hydroxysuccinimide (NHS) esters, isothiocyanates, and activated carboxyl groups, enabling efficient coupling to fluorophores, peptides, proteins, antibodies, polymers, and nanoparticles under mild aqueous conditions. Because amine-reactive labeling reagents are broadly available, amino-modified oligonucleotides remain one of the most flexible platforms for probe labeling, capture probe immobilization, and oligonucleotide-based biosensor construction.
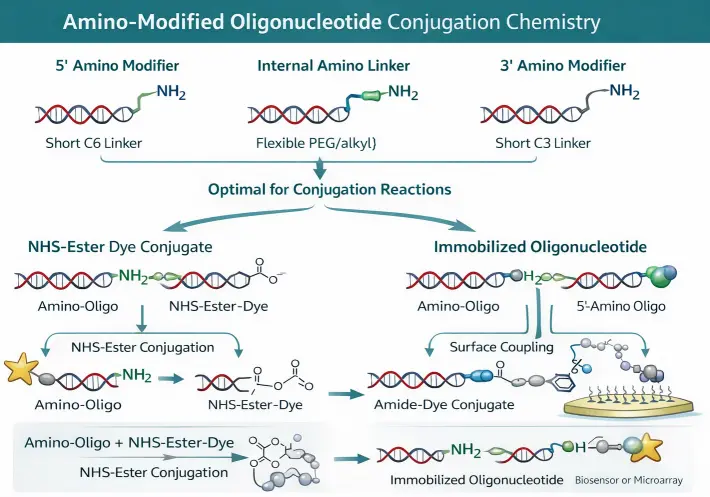
Amino-Modified Oligonucleotide Architecture. Representative terminal and internal amine installations used for post-synthetic labeling, surface coupling, and bioconjugation workflows.
ISO 9001:2015 /
ISO 13485:2016
45+ Years of Expertise
U.S.A. Facilities-Texas
GLP/GMP-Aligned
5′ / 3′ / Internal Amino
DNA & RNA Compatible
Linker and Nucleobase Options
NHS Dye Conjugation Ready
Surface Immobilization Compatible
Design insight: the optimal amino modifier depends on conjugation chemistry, desired position, linker length, steric accessibility, nucleic acid type, and the size of the intended labeling partner or immobilization surface.