Photo-regulated oligonucleotides contain photolabile protecting groups or photo-responsive structural elements that undergo wavelength-dependent photochemical activation. These chemistries enable precise control over activation kinetics, spatial localization, and functional state, allowing oligonucleotides to be selectively activated, cleaved, or structurally modulated for controlled experiments in molecular biology, cellular imaging, synthetic biology, and nucleic acid nanotechnology.
These systems are widely used in optochemical biology, light-controlled gene expression, and dynamic nucleic acid nanostructures, where external optical triggers replace chemical or enzymatic activation.
Photoregulation efficiency depends on photolysis quantum yield, steric blocking efficiency, and local duplex destabilization effects introduced by the modification, all of which influence activation kinetics and functional recovery.
Two major classes dominate this area. Photocaged oligonucleotides use light-removable protecting groups to block hybridization, enzymatic recognition, or functional activity until illumination removes the cage. Photo-cleavable oligonucleotide systems use light-sensitive linkers, spacers, or terminal handles that break upon illumination to release cargo, restore activity, or trigger structural rearrangement.
These strategies are especially useful for spatiotemporal control of gene regulation, triggered molecular release, light-activated affinity workflows, optochemical biology, and dynamic DNA nanostructure control. Depending on the photochemical group, activation may occur under UV light or, in selected systems, under longer visible wavelengths that may be more biologically compatible.
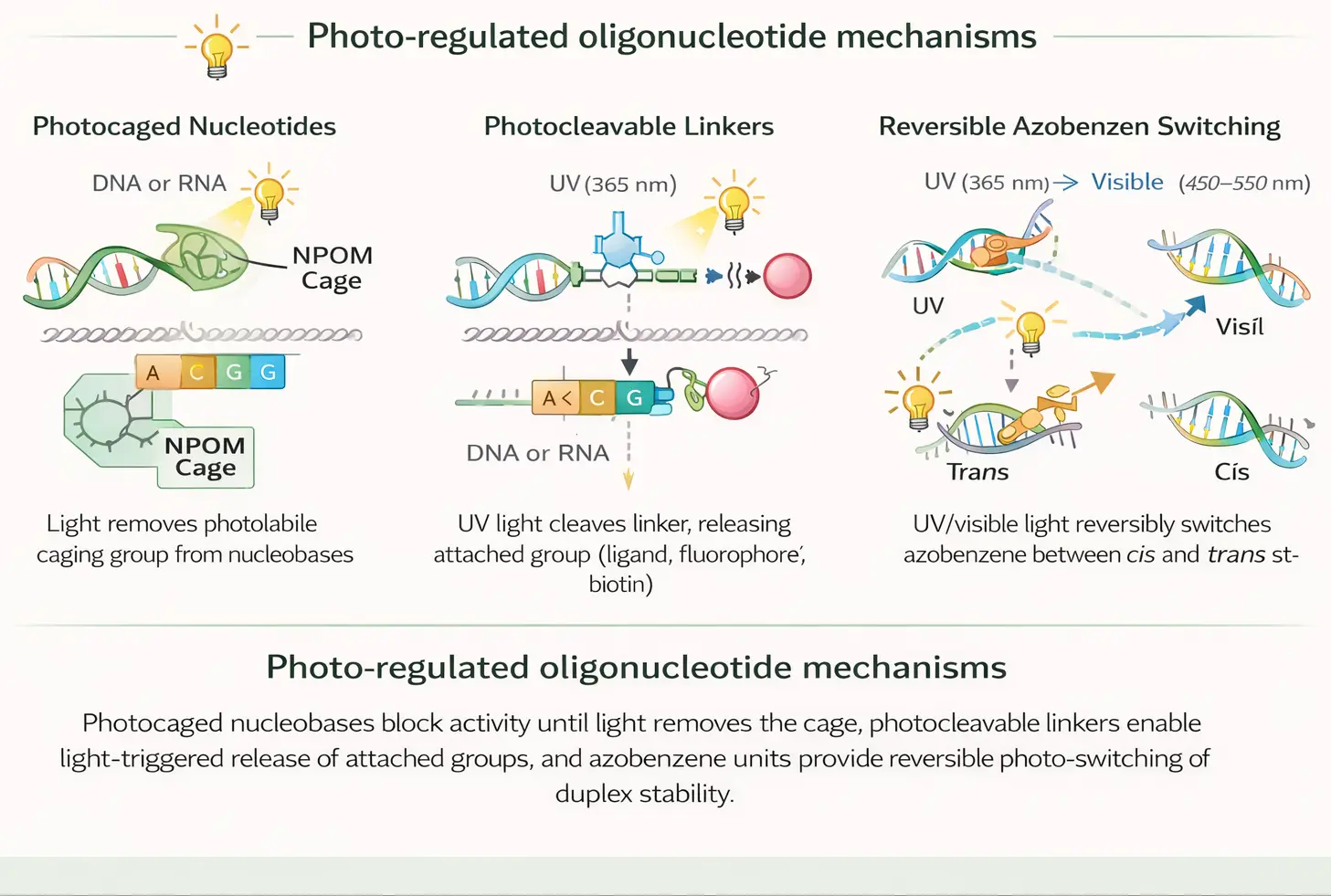 Photo-regulated oligonucleotide mechanisms. Photocaged nucleobases block activity until light removes the cage, photocleavable linkers enable light-triggered release of attached groups, and azobenzene units provide reversible photo-switching of duplex stability.
Photo-regulated oligonucleotide mechanisms. Photocaged nucleobases block activity until light removes the cage, photocleavable linkers enable light-triggered release of attached groups, and azobenzene units provide reversible photo-switching of duplex stability.
Photo-regulated oligonucleotide chemistry enables researchers to use light as a non-contact external trigger for activation, release, switching, or structural control of DNA and RNA systems.