Since 1984, Bio-Synthesis has specialized in peptide synthesis for biotechnology, pharmaceutical, diagnostic, and academic research markets. Our deep technical expertise and advanced peptide synthesis platforms allow us to consistently deliver high-purity peptides with exceptional sequence accuracy and reproducibility.
This regulatory framework ensures that every peptide synthesis project meets strict quality standards suitable for research and diagnostic development.
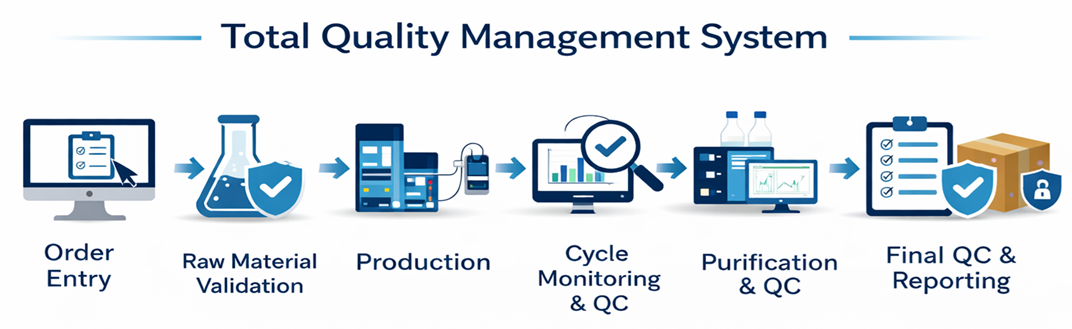
Delivering high-quality peptide synthesis and oligonucleotides begins with a robust, fully integrated quality control and management system.
At Bio-Synthesis, our proprietary software platform manages the entire workflow — from order entry and production tracking to final product release and dispatch. This end-to-end system ensures that quality is continuously monitored, documented, and maintained at every stage, providing our customers with confidence, consistency, and reliability.
With over 45 years of expertise in peptide synthesis, Bio-Synthesis is uniquely equipped to handle even the most technically demanding custom biomolecules. Our team of experienced Chemists is here to support you at every stage—offering guidance in experimental design, application optimization, and technical troubleshooting. When you work with us, you're not just getting a product—you're gaining a partner in scientific success.
At Bio-Synthesis, our unwavering commitment to quality is reflected in every oligonucleotide we produce. We follow stringent quality assurance protocols to ensure reliability, consistency, and performance:
With over four decades of manufacturing excellence, we stand behind our promise of high-quality, application-ready oligonucleotides.
Learn more about our Custom Process Developments
To support your regulatory and research needs, Bio-Synthesis provides comprehensive quality control documentation, including:
Our quality documentation ensures transparency, traceability, and confidence in every batch delivered. Learn more about our Peptide analytical services
At Bio-Synthesis, our mid-scale peptide manufacturing process is designed to meet rigorous quality and regulatory standards. For all projects in the 1 to 100 g range, we implement the following practices:
Our robust mid to large scale synthesis platform ensures high-quality, reproducible results—ready for downstream development.

Our state-of-the-art facility houses over 50 synthesizers, supporting the full spectrum of peptide production—from high-throughput small-scale plate synthesis to midscale and large-scale manufacturing—all under one roof.
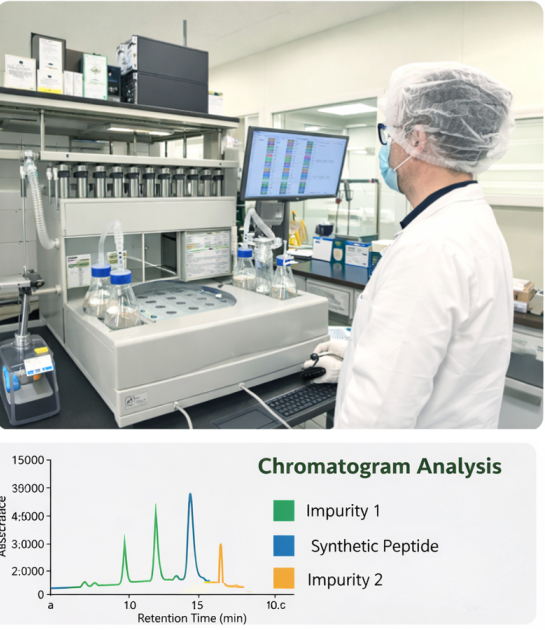


High-resolution LC–MS instrumentation for identity confirmation and impurity profiling.



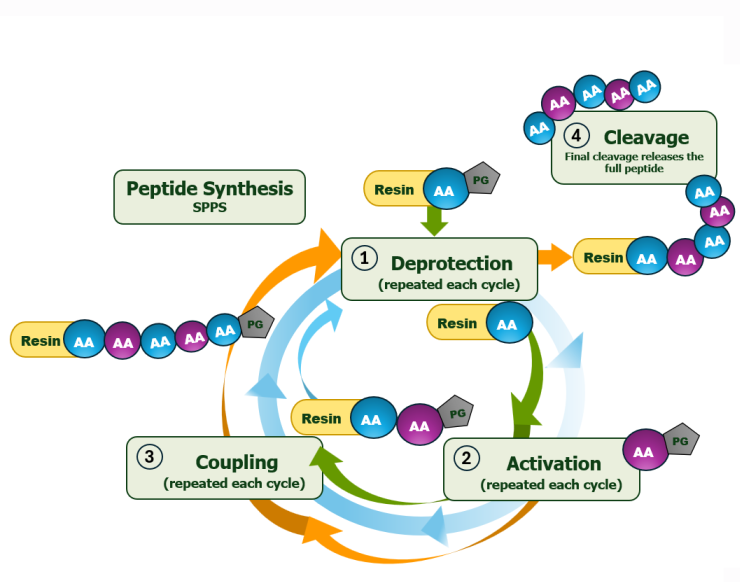
At Bio-Synthesis, purification is a critical component of our peptide manufacturing process—ensuring optimal yield and exceptional purity. Our experienced chromatographers and process chemists utilize advanced high-pressure liquid chromatography (HPLC) techniques to consistently deliver high-quality peptdes. We provide end-to-end, scientist-to-scientist collaboration—from project planning to final delivery—supporting quantities ranging from micrograms to hundreds of grams. This commitment reflects our customer-first philosophy and dedication to excellence.
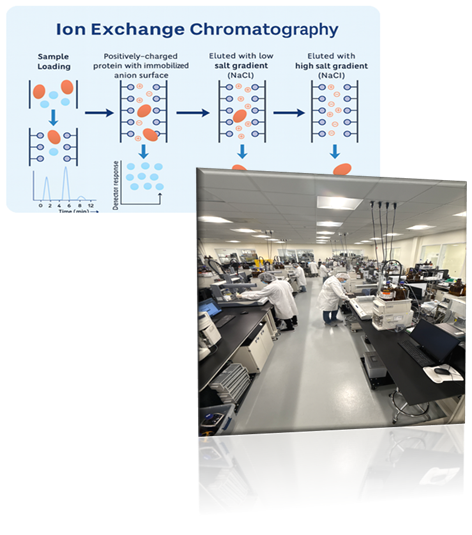
We offer industry-standard purification methods, including HPLC, reverse-phase, and ion-exchange chromatography. Additionally, our team can develop customized in-house protocols tailored to your specific yield and purity requirements.

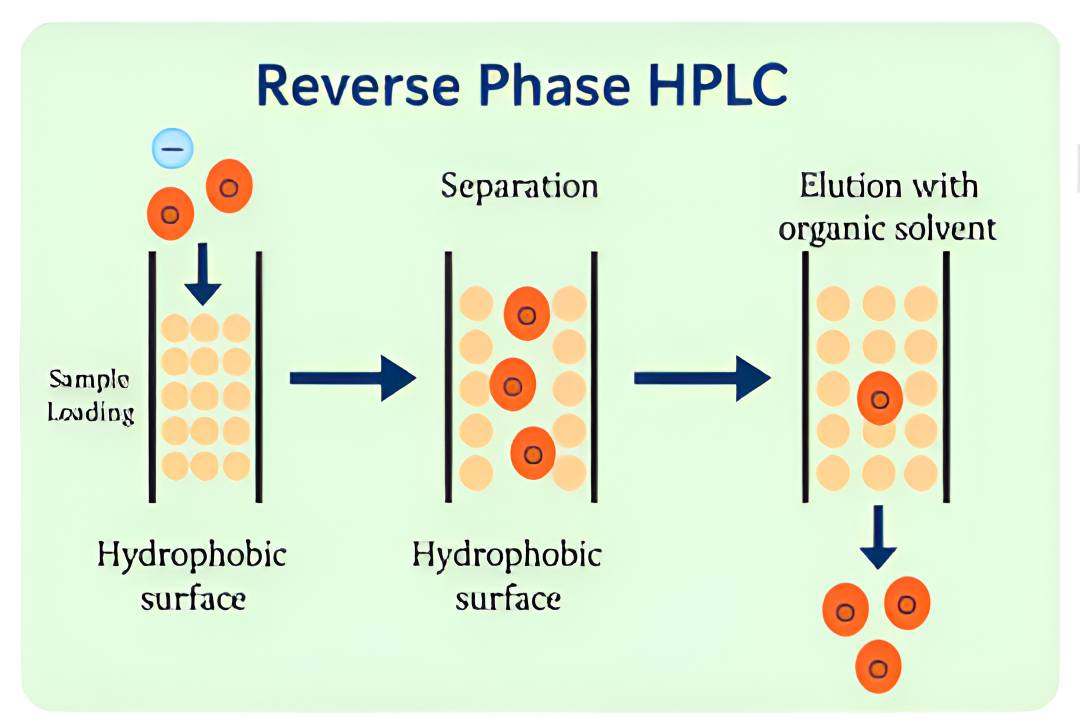
CLick here for additional information on Peptide Release QC Services
At Bio-Synthesis, the quality of your custom oligonucleotides is our highest priority. We provide comprehensive release testing services that align with the most stringent industry and regulatory standards. Whether your project requires non-GMP or cGMP-compliant testing, our experienced team ensures each oligo meets all critical specifications—guaranteeing reliable performance, regulatory compliance, and batch-to-batch consistency.
At Bio-Synthesis, we understand that every product is unique—and so are its analytical requirements. Our Custom Analytical Method Development and Validation services are purpose-built to support your therapeutic or diagnostic programs with the rigorous, regulatory-aligned testing needed to ensure product quality, consistency, and compliance at every stage of development.
Whether you're in early research, preclinical testing, or preparing for GMP production, we provide comprehensive analytical solutions tailored to your project’s specific molecular design, formulation, and regulatory pathway. From fit-for-purpose method development to full method validation, our team of scientists delivers results you can rely on—efficiently and with full transparency.
Our method development process is designed to accommodate the unique structural and functional characteristics of peptides, oligonucleotides, peptide bioconjguation, and other complex biomolecules. Services include:
Every method we develop is optimized for sensitivity, specificity, and reproducibility, ensuring accurate characterization of your product under real-world production and storage conditions.


Validation is conducted in accordance with ICH Q2(R1), USP, and FDA/EMA guidelines to ensure your analytical procedures meet all regulatory expectations for accuracy, precision, linearity, range, specificity, and robustness.
Our validation services cover: Full and partial method validation (for RUO, GLP, or GMP applications) System suitability testing Forced degradation and stress testing studies Analytical protocols and reporting documentation to support IND, BLA, or NDA submissions Method bridging and cross-site validation support Whether you're developing custom oligos for therapeutic use or producing GMP-grade diagnostics, we help ensure that your analytical data stands up to regulatory scrutiny—every time.
As part of our broader manufacturing and quality platform, our analytical services are fully integrated into our Total Quality Management (TQM) system. This ensures traceability, data integrity, and consistency across all batches and all phases—from small-scale pilot runs to full GMP production.
At Bio-Synthesis , we are dedicated to delivering excellence through the following core principles:
We strictly adhere to all applicable customer, quality, safety, and environmental requirements to ensure regulatory integrity and client trust.
We actively monitor customer satisfaction, respond to feedback, and implement necessary improvements to consistently exceed expectations.
We drive continual improvement across our processees, people, and systemss by setting and reviewing measurrable Quality Objectives. ensuring the ongoing effectiveness of ourr Quality Management System (QMS)
Trusted by biotech leaders worldwide for over 45+ years of delivering high quality, fast and scalable synthetic biology solutions.