Click chemistry is widely used for post-synthetic oligonucleotide conjugation because it is highly selective, compatible with aqueous conditions, and can form stable triazole-linked products under mild reaction conditions [1].
Common click-compatible handles include azide, alkyne, 5′-Hexynyl, Octadiynyl-dU, DBCO, and related strained alkyne systems...
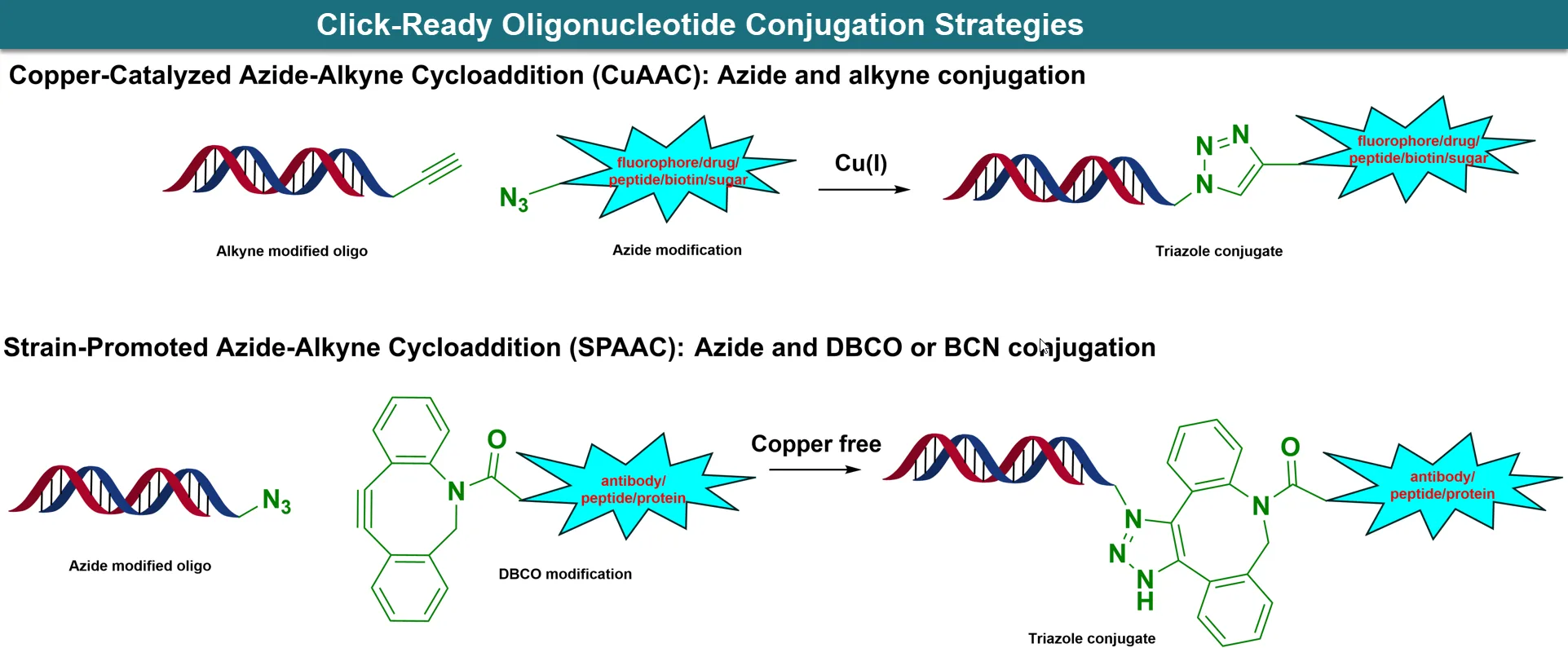
Click-Ready Oligonucleotide Conjugation Architecture. Representative azide, alkyne, 5′-Hexynyl, Octadiynyl-dU, and DBCO-based oligo handles used for CuAAC and copper-free SPAAC conjugation to fluorophores, peptides, proteins, polymers, and nanoparticles.