Bio-Synthesis provides custom fluorescent labeling of oligonucleotides for molecular diagnostics, quantitative PCR, fluorescence in situ hybridization (FISH), imaging, and multiplex detection assays. A broad range of fluorophores is available, including standard qPCR reporters, high-brightness imaging dyes, far-red and near-infrared labels for reduced autofluorescence, and dark quenchers used in dual-labeled probes, molecular beacons, and real-time detection systems.
Fluorescent labeled oligonucleotides are custom DNA or RNA constructs modified with reporter dyes for nucleic acid detection, molecular diagnostics, imaging, hybridization, and quantitative PCR workflows. Fluorophores can be introduced during solid-phase oligonucleotide synthesis using dye phosphoramidites or installed after synthesis through reactive handles such as amino, thiol, azide, alkyne, or DBCO groups, enabling flexible probe design for qPCR, FISH, molecular beacons, microarrays, sequencing, and advanced fluorescence-based assays [1–3].
Compared with unlabeled oligos, fluorescently modified oligonucleotides provide direct optical readout, multiplexing potential, and compatibility with quencher-based signal control. Careful selection of dye class, excitation/emission spacing, labeling position, spacer chemistry, and purification strategy is important for achieving strong signal-to-noise performance and reproducible assay behavior, especially in multiplex probe systems and imaging applications [4–6].
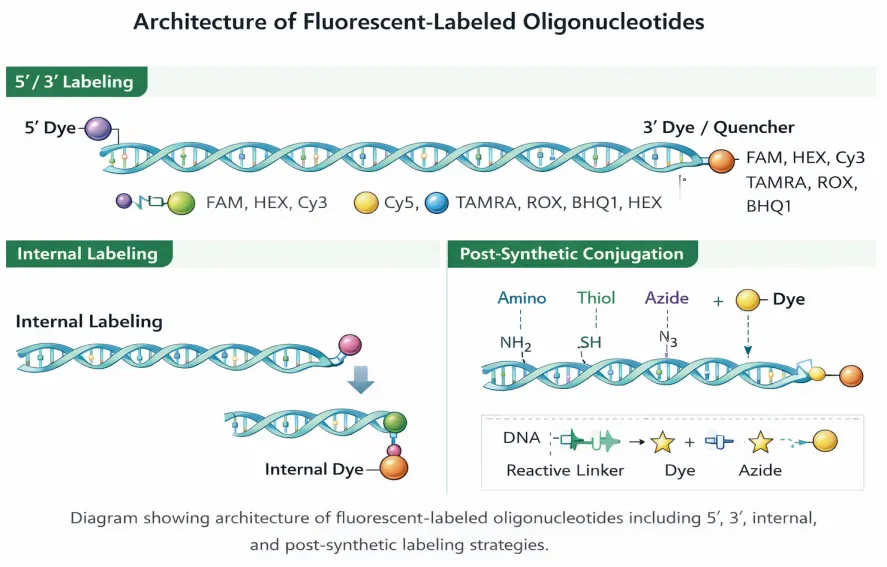 Architecture of Fluorescent-Labeled Oligonucleotides.
Representative 5′ labeling, 3′ dye or quencher labeling, internal labeling, and post-synthetic conjugation strategies used for fluorescent probe and assay design.
Architecture of Fluorescent-Labeled Oligonucleotides.
Representative 5′ labeling, 3′ dye or quencher labeling, internal labeling, and post-synthetic conjugation strategies used for fluorescent probe and assay design.
ISO 9001:2015 /
ISO 13485:2016
45+ Years of Expertise
U.S.A. Facilities-Texas
GLP/GMP-Aligned
5′ / 3′ / Internal Labeling
Direct Amidite or Post-Synthetic
HPLC purified
DNA & RNA Compatible
200+ Dye Options
Design insight: the best fluorescent labeling strategy depends on the assay format, optical channel plan, dye–quencher pairing, labeling position, and whether the fluorophore is better introduced during synthesis or through post-synthetic conjugation.